INTRODUCTION
myo-Inositol is a cyclic alcohol derivative from glucose that provides the backbone for phytate. Phytic acid is the primary form of phosphorus in seeds, the common name for
myo inositol-1,2,3,4,5,6-hexa
kisphosphate (IP6), and can account for up to 75% of total P present in seeds [
1]. There are 2 conformational forms of phytic acid, 5ax/1eq and 1ax/5eq, with the latter being the sterically favorable form containing 1 axial phosphate and 5 equatorial phosphates. Form 5ax/1eq is sterically hindered, generally referred to as phytin, and can be stabilized upon formation of salt complexes with metal cations [
2]. These complexes reduce the bioavailability of P and other essential mineral cations for monogastrics, due to low levels of endogenous phytase present in seeds as well as in the gastro-intestinal tract of monogastrics. The hexa
kisphosphoric acid ester of
myo-inositol or phytate is another common structure of phytic acid.
myo-Inositol-hexa
kisphosphate phosphohydrolase (EC 3.1.3.8) is the endogenous seed enzyme responsible for phytate degradation during germination to release phosphorus and other cation metals. Thus, animal diets containing seeds as dietary ingredients will have low level of endogenous phytase activity, because vertebrates’ low secretion of endogenous phytase and low endogenous phytase activity in grains and grain byproducts result in low standardized total tract digestibility (STTD) of P present in vegetable feedstuffs [
3]. Grain byproducts obtained from fermentation processes, such as distillers dried grains with solubles (DDGS), have considerably higher STTD of P [
3,
4] compared with corn.
The efficacy of fungal- and bacterial-derived phytases have been extensively researched over the last decades in complete diets [
5], but few studies have focused on its P releasing effects on individual ingredients [
4,
6,
7]. Previous results indicate that the response of phytase supplementation on bone ash [
8] or STTD of P in diets [
9] can be explained by fitting either quadratic or broken-line curves to determine the inflexion or break-point, both of which correspond to the maximal concentration of phytase needed to reach a plateau in the response variable. Therefore, the objectives of this work were to quantify as well as to fit linear, quadratic, exponential or broken-line curves for STTD of P in individual ingredients commonly used in swine diets with increasing concentrations of a novel bacterial-derived 6 phytase supplementation.
RESULTS AND DISCUSSION
The analyzed concentration of total P in the tested feed ingredients is presented in
Table 2. Analyzed results are in close agreement with previously reported values and were determined to be within±1 standard deviation [
3]. Exogenous phytases have been successfully used in the swine and poultry industries for many decades for the improvement of P utilization [
5]. Phytases are generally produced from microbes or they may originate from plants [
9]. It is well-known, however, that phytases originating from different organisms exert different physical and biochemical properties, which dictate the efficacy of exogenous phytases in swine and poultry [
20]. Thus, as new sources of phytase are developed, evaluation of its efficacy in improving P utilization in monogastric animals is warranted. The phytase used in the present study is a novel bacterial-derived 6-phytase expressed in
Psedomonas fluorescens and to the best of our knowledge, no data is available demonstrating its efficacy in improving P digestibility in feed ingredients commonly fed to pigs.
Values for the STTD of P determined for each tested feed ingredient without the addition of phytase are in agreement with values reported in the literature ([
3,
21];
Table 2). For all feed ingredients tested in this experiment, the STTD of P increased with graded concentrations of phytase (
Table 3). Using BIC, it was determined the best model that describes the relationship between phytase dose supplementation and STTD of P was broken-line for corn, exponential for CM, RB, SBM, SFM, and wheat, and linear and exponential were equally good for DDGS and sorghum (
Table 4). The concentration of phytase needed to maximize the STTD of P for both DDGS and sorghum was estimated to be greater than 2,000 FTU/kg, which is outside the studied range for the enzyme, and hence it is concluded that a linear model is the best fit (
Table 5). Quadratic regression had the highest BIC value for all ingredients indicating the poorest goodness of fit (
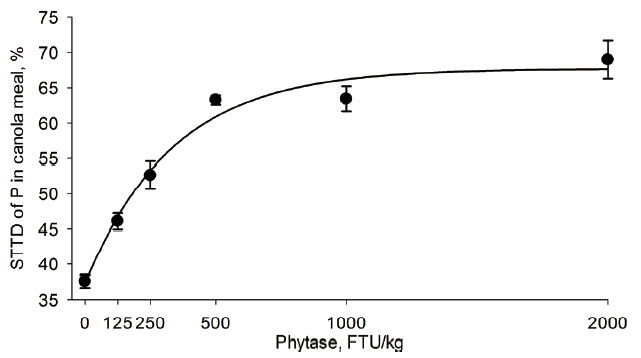
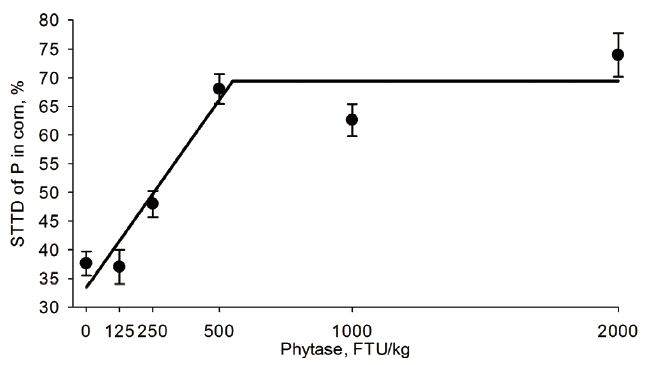
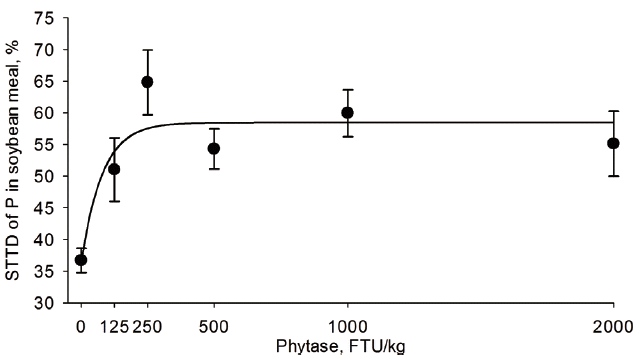
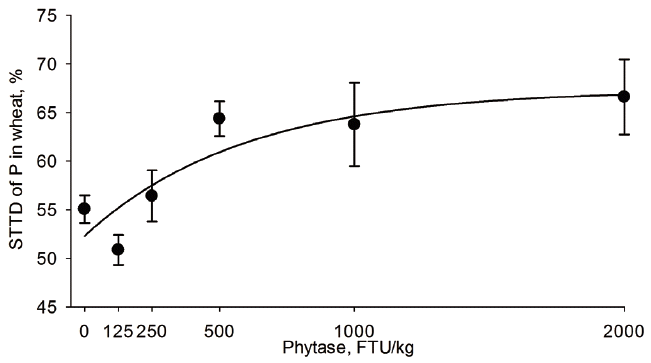
Table 4). The STTD of P increased from 37.6% to 69.0% in CM, 37.6% to 73.9% in corn, 68.6% to 77.0% in DDGS, 10.3% to 33.1% in RB, 41.2% to 72.7% in sorghum, 36.7% to 55.1% in SBM, 26.2% to 59.6% in SFM, and 55.1% to 66.6% in wheat when phytase was supplemented at 0 or 2,000 FTU/kg, respectively (
Table 3). Almeida and Stein [
11] reported linear and quadratic response for the STTD of P in corn when phytase was supplemented from 0 to 1,100 FTU/kg. In the present study, STTD of P increased with graded concentrations of phytase in corn in a broken-line manner (i.e., lowest BIC score). Thus, both studies indicate that the amount of P released from corn depends on the amount of phytase included in the diet. However, the ability of different phytases to increase STTD of P and best regression model to describe each enzyme response in different ingredients needs to be evaluated simultaneously
in vivo. The response measured for the STTD of P in DDGS in the present experiment; however, does not agree with previously reported data [
11], in which phytase did not improve the STTD of P in DDGS. Importantly, the STTD of P in DDGS reported by [
11] was 76.9%, which is higher than the determined STTD of P for DDGS in this experiment (68.6%). Thus, it is possible that phytase responded differently in this experiment because of a higher concentration of phytate in DDGS used herein, although the present experiment did not measure phytate concentration or due to differences in the ability of phytases to hydrolyze phytate in DDGS.
The phytase concentration (FTU/kg) needed to maximize the STTD of P in the tested feed ingredients determined by the best regression model (
Table 4) is shown in
Figures 1 to
6 for CM, corn, RB, SBM, SFM, and wheat. When determined by broken line analysis, the estimated minimum phytase dose (FTU/kg) needed to plateau STTD of P was, respectively, 454 for 65.2% in CM, 550 for 69.4 in corn, 194 for 58.9% in SBM, 785 for 58.9% in SFM, and 603 for 65.3% in wheat. The break-point for RB, sorghum, and DDGS was determined to be 2,000 FTU/kg indicating that a plateau was not obtained. The quadratic regressions indicate that the phytase dose (FTU/kg) needed for highest STTD of P was, respectively, 1,494 for 71.6% in CM, 1,547 for 74.5% corn, 732 for 65.4% in DDGS, 1,968 for 73.2% in sorghum, 1,153 for 63.8% in SBM, 1,530 for 62.7% in SFM, and 1,565 for 67.6% in wheat; estimates for RB were outside the studied range. Using an exponential model, the calculated phytase dose (FTU/kg) needed for highest STTD of P defined as 95% of the upper asymptote [
18] was, respectively, 735 for 64.3% in CM, 1,112 for 70.0% in corn, 160 for 55.5% in SBM, 1,219 for 57.8% in SFM, and 881 for 64.0% in wheat; estimates for DDGS, sorghum, and RB were outside the evaluated range indicating the enzyme follows a fairly linear response within the range studied for these three ingredients.
The broken-line analysis has been used elsewhere to determine the phytase concentration needed to maximize the STTD of P in complete diets [
9]. To the best of our knowledge, this approach has not been used for individual feed ingredients in pigs or other species. Under other scenarios (e.g., determination of AA requirements) the robustness of estimates based on broken line analysis has been questioned, as this analysis tends to underestimate the requirement for a significant portion of the population [
22,
23]. The reason for this is because the broken line regression estimates the breakpoint at an average for the test population [
24]. Conversely, the quadratic model is believed to overestimate the requirement for the majority of the sample population [
24]. It has been previously reported that linear and nonlinear models (e.g., exponential) used to determine nutrient requirements using growth data results in similar estimates when adequate fits are obtained for both models [
25]. Nonetheless, the better goodness of fit of the exponential model makes it a better approach to describe and interpret the data [
25]. These concerns are obvious in the present study, in which there are differences among the phytase dose (FTU/kg) needed to maximize the STTD of P using different approaches (e.g., 550 for broken line, 1,547 for quadratic, and 1,125 for exponential in corn). However, for other ingredients like SBM the estimated phytase dose (FTU/kg) needed to maximize the STTD of P is similar between broken-line and exponential models (194 vs 160, respectively). Thus, to mitigate some of the shortcomings of models when estimating the optimum concentration for a given response criterion, a combination of model approach has been proposed [
22,
23]. In these analyses, the requirement for a given response variable is defined as the first intercept of the quadratic or exponential regression with the plateau of the broken-line. The first intercept between the plateau of the broken line with the quadratic and exponential models is commonly used as conciliatory values among models and those estimates are presented in
Table 5. It should be noted again that the quadratic model had the highest BIC score for all ingredients indicating the poorest goodness of fit for this regression approach. Thus, the applicability of the intercept of the plateau of the broken-line model with the quadratic model could be debatable. Although the aforementioned approaches serve to indicate the maximum STTD of P achieved in each feed ingredient by phytase supplementation and also illustrate the fact that phytase efficacy differs among different feed ingredients with a given phytase dose, the application of such information in commercial diets is cumbersome. In practical formulation, nutritionists use a given dose of phytase, which as demonstrated here, may not necessarily maximize the STTD of P in all ingredients used in the complete feed. Nevertheless, the regression models developed here may be used to predict the amount of STTD P released by this novel phytase for any complete feed where the ingredients tested here are used. Each ingredient, however, will have a different equation which in some cases could be better described by broken-line, linear, or exponential models according to the lowest BIC values among models (
Table 5).
The results from this study indicate that the efficiency of phytase in improving the STTD of P varies among feed ingredients and this might be attributed, in part, to composition, level, and location of phytate within different plant-based feed ingredients [
5]. Most of the phytate in corn is located within the germ, whereas in wheat and sorghum phytate is mainly found within the aleurone layers [
26,
27]. Thus, it is possible that phytate in corn is more easily accessible to exogenous phytases than phytate located within the fibrous matrix of wheat and sorghum. In fact, it was determined that IP6 ileal digestibility in broilers was greater in corn-based diets (51.5%) than in wheat and sorghum-based diets (49.9% and 34.0%, respectively; [
28]). In another study with broilers, P released from IP6 with 100 FTU/kg of exogenous phytase in corn was 4.66% compared with 2.69% in wheat [
29]. These reports are in agreement with present results, the estimated dose of phytase (FTU/kg) needed to maximize the STTD of P was 550 for corn, 881 for wheat, and the estimated value for sorghum was greater than the maximum tested dose of 2,000 FTU/kg of phytase.
In non cereal seeds such as soybean, canola, and sunflower, most of phytate is present within the protein matrix, but in the case of SBM, phytate is homogenously distributed within the seed, whereas in other oilseeds it accumulates in specific sites [
30]. Thus, as determined in this study, it is possible that different responses measured for each oilseed could have been a result of the different interactions between phytate and other nutrients (e.g., protein) in each ingredient [
30].
Because phytase supplementation increases the STTD of P differently depending on the feed ingredient, it seems logical that the response to phytase supplementation also varies depending on diet composition. Three plausible diets were formulated and corroborate with this hypothesis (
Figure 7). For example, the basal STTD P for plausible Diet A was calculated to be 0.166% and adding 500 FTU/kg of the novel phytase evaluated in this study should increase STTD P to 0.287% (
Table 6), meaning that the STTD P value for this phytase in this diet will be 0.12%. Similar calculations for diet B and diet C indicate a STTD P value of 0.091% and 0.086%, respectively. The STTD P value of the enzyme was higher in complex diets that contained SFM and CM than in the simpler corn-SBM diet. Interestingly, in the diets that contained RB, the predicted improvements in STTD P value continue to increase with dose, whereas the other diets did reach a plateau at 750 and 1,000 FTU/kg of phytase providing a maximum of 0.145% and 0.091% STTD P in diet A and diet B, respectively. The implication of our findings and this simulation is that nutritionists should refrain from using a fixed P matrix value indiscriminately as the value of any given phytase will likely depend on the selection of ingredients and their inclusion concentrations within a diet.
In conclusion, our results demonstrate the effectiveness of this novel microbial 6-phytase expressed in Psedomonas fluorescens to improve the STTD of P in various feed ingredients commonly used in swine diets. The efficiency of this novel enzyme to hydrolyze phytate is best described with a broken-line model for corn, an exponential model for CM, SBM, SFM, and wheat, and a linear model for DDGS, RB, and sorghum for the phytase evaluated in the present study within the range of 0 to 2,000 FTU/kg. The concentration of phytase needed to maximize the STTD of P varied for different feed ingredients and this needs to be taken into consideration when determining the matrix values for phytases, depending on the type of ingredients used when mixing complete diets for pigs.










 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print





