Effect of High Dietary Carbohydrate on the Growth Performance, Blood Chemistry, Hepatic Enzyme Activities and Growth Hormone Gene Expression of Wuchang Bream (Megalobrama amblycephala) at Two Temperatures
Article information
Abstract
The effects of high carbohydrate diet on growth, serum physiological response, and hepatic heat shock protein 70 expression in Wuchang bream were determined at 25°C and 30°C. At each temperature, the fish fed the control diet (31% CHO) had significantly higher weight gain, specific growth rate, protein efficiency ratio and hepatic glucose-6-phosphatase activities, lower feed conversion ratio and hepatosomatic index (HSI), whole crude lipid, serum glucose, hepatic glucokinase (GK) activity than those fed the high-carbohydrate diet (47% CHO) (p<0.05). The fish reared at 25°C had significantly higher whole body crude protein and ash, serum cholesterol and triglyceride, hepatic G-6-Pase activity, lower glycogen content and relative levels of hepatic growth hormone (GH) gene expression than those reared at 30°C (p<0.05). Significant interaction between temperature and diet was found for HSI, condition factor, hepatic GK activity and the relative levels of hepatic GH gene expression (p<0.05).
INTRODUCTION
The carbohydrate utilization of fish varies between fish species and carbohydrate sources (Wilson, 1994; Krogdahl et al., 2005). Fish are known to have a limited ability for digestion and metabolism of carbohydrate and hence, excessive intake of this nutrient may result in nutritional problems (Jauncey, 1982; Roberts, 1989; Lall, 1991). Excess carbohydrates reduce the growth rate and are often accompanied by poor feed utilization (Hemre et al., 2002). Fish species are poikilothermic and their feed intake, growth and metabolic responses are affected by surrounding temperature (Peres and Oliva-Teles, 1999). Temperature is one of the key factors influencing the physiological characteristics of ectotherms (Jobling, 1994; Gillooly et al., 2001; Hochachka and Somero, 2002; Clarke, 2004), and it also modulates utilization of nutrients by fish (Keembiyehetty and Wilson, 1998). Brett et al. (1969) reported that fish convert their food to energy more efficiently near their optimal temperature. Increasing water temperature improved starch digestibility (Médale et al., 1999) while enzyme activities were higher at lower temperatures, denoting thermal compensation for acclimation to low temperature in carp (Shikata et al., 1995). Besides growth performance, temperature was shown to affect feed efficiency, protein utilization, body composition and glycolytic, lipogenic, and gluconeogenic capacities in European sea bass (Person-Le Ruyet et al., 2004; Enes et al., 2006; Moreira et al., 2008).
Wuchang bream (Megalobrama amblycephala) is a Chinese freshwater herbivorous species (Ke, 1975) with high potential for aquaculture. Its optimum temperature is about 25°C (Ke, 1986). Due to the merits of this species including tender flesh, fast growth, economic profitability and cultural values (Zhou et al., 2008), Wuchang bream is widely cultured in China with the output of 625,789 tons in 2009—an increase of 31.5% in the past decade. This species has been introduced to North America (northern Canada to southern Mexico), Africa, Europe, and other Asian countries (Ke, 1986).
Recent studies showed that capacity for carbohydrate utilization of Wuchang bream was reported to be between 25% and 34% (Yang et al., 1989; Zhou et al., 2013). However, the effect of high dietary CHO level on the growth and physiological responses of Wuchang bream reared at different temperatures are not known. Thus, the objectives of this study are to examine whether the effects of high dietary CHO on the growth performance, blood chemistry, hepatic enzyme activities and the expression of hepatic growth hormone (GH) in Wuchang bream at different water temperatures and to try to identify the molecular mechanisms for these effects.
MATERIALS AND METHODS
Fish and diets
Juvenile Wuchang bream, M. amblycephala (15.73±0.03 g), were obtained by fish farm of Freshwater Fisheries Research Center, Chinese Academy of Fishery Sciences in China. Fish were acclimated at either 25±0.5°C or 30±0.5°C in cylindrical fibreglass tanks (300 L) in a re-circulating system for 15 days before the feeding experiment. During the period, fish were fed to apparent satiation with a control diet every day. The water oxygen content was approximately 6.0 mg/L during the experimental period. A 12 L:12 D photoperiod was used.
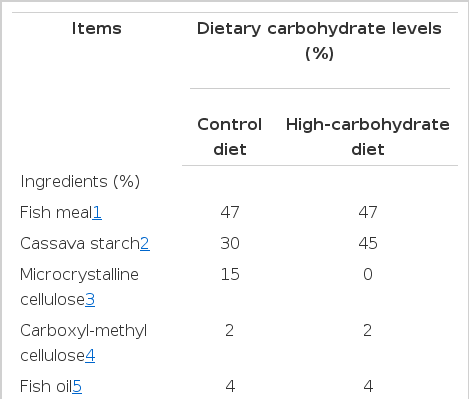
Using fishmeal as protein source, fish oil as lipid source and cassava starch as carbohydrate source, two isonitrogenous (34% crude protein) and isolipidic (8.1% crude lipid) semi-purified diets were formulated to contain 31% and 47% carbohydrate, and these were referred to as a control and a high carbohydrate level respectively (Table 1). Dry ingredients were mixed thoroughly in a mixer, then water was added and mixed. Two-mm-diameter pellets were wet-extruded. The pellets were dried in a forced air oven at 40°C to a moisture content of 10% and then stored at −20°C until use.
Rearing management
At the start of the experiment, the fish were fasted for 24 h and then weighed. Nine fish from each temperature were sampled. The fish from each temperature were then divided into two groups, each group comprising three tanks of 20 fish, and fish fed to satiation with the control and high-carbohydrate diets three times (06:30, 11:30, and 16:30 hours respectively) daily for 8 weeks. Water was oxygenated at all times using an aerator. Feces and debris were removed daily using a siphon. Water temperature was measured every day, and water quality was measured every week. During this period, feed consumption was recorded daily. The number and weight of the dead fish was also recorded. During the test period, the water quality on average was as follows: dissolved oxygen (DO)>6 mg/L, NH3<0.05 mg/L, H2S<0.1 mg/L, and pH 6.8 to 7.3. After the completion of the test period (70 days), serum and liver samples were collected. At the end of the feeding trial, the fish were fasted for 24 h, and then counted and weighed. The serum and liver samples were collected.
Serum and liver sample collection
At the end of the growth trial, the fish were starved for 24 h and weighed. Nine individuals from each group (3 tanks, 3 fish per tank) were anesthetized with MS-222 and sampled. Blood was sampled from the caudal vein, centrifuged at 3,000 g for 10 min (4°C), and kept frozen until analysis. The liver was excised, frozen in liquid nitrogen and then stored at −80°C until assay.
Proximate analysis
The contents of dry matter, crude protein, crude lipid, and ash were determined for the experimental diets and the fish bodies. Chemical analyses were conducted using the following AOAC (1997) procedure: dry matter by drying in an oven at 105°C to a constant weight; crude protein (nitrogen×6.25) using the Kjeldahl method after acid digestion (FOSS KT260, Höganäs, Sweden); crude lipid by ether-extraction in a Soxtec System HT (Soxtec System HT6, Tecator, Höganäs, Sweden); and ash by incineration in a muffle furnace at 550°C for 4 h. Carbohydrate contents of feed were analyzed by the 3,5-dinitro salicylic acid method (Yu et al., 1997). The hepatic glycogen content was determined as described in Plummer (1987).
Plasma glucose, triglyceride, and cholesterol levels
Plasma glucose, triglyceride, and cholesterol content were measured using the glucose oxidase method, the enzymatic (glycerol phosphate oxidase) and colorimetric (PAP) method, and the enzymatic (cholesterol oxidase) and colorimetric method, respectively, using test kits purchased from Junshi Biotechnology Co., Ltd. (Shanghai, China).
Enzyme activity analysis
A frozen sample of liver was homogenized (dilution 1/10) in ice-cold buffer consisting of 80 mM Tris (pH 7.5), 5 mM ethylene diamine tetraacetic acid (EDTA), 1 mM KH2PO4, 2 mM NaHCO3 and 1.4 mM dithiothreitol. The homogenate was centrifuged at 4,000 rpm for 10 min at 4°C (Cai, 2004), and then the supernatant was centrifuged at 12,000 rpm for 20 min at 4°C. The supernatant was separated and divided into three parts for measurement of glucokinase (GK), phosphoenolpyruvate carboxykinase (PEPCK), and glucose-6-phosphatase (G6Pase) activity.
Glucokinase (EC2.7.1.2) activity was measured using the glucose-6-phosphate dehydrogenase (G6PDH) coupling method (Tranulis et al., 1996; Panserat et al., 2000a). The reaction system contained 65 mU/mL G6PDH, 2 mM NADP+ (oxidized form of nicotinamide-adenine dinucleotide phosphate) 7 mM adenosine triphosphate, 80 mM Tris, 5 mM EDTA, 8 mM MgSO4, 1 mM KH2PO4, 2 mM NaHCO3, 0.2 mM dithiothreitol, and 100 mM glucose at pH 8.2. The glucose concentration was 0.64 mM to 1,277 mM in the kinetic study. This assay for measuring GK activity using frozen samples necessitated correction by measuring glucose dehydrogenase activity (E.C. 1.1.1.47) as described previously (Tranulis et al., 1996). One unit of GK activity was defined as the amount of NADPH (reduced form of nicotinamide-adenine dinucleotide phosphate) generated by per g protein per minute at 30°C.
Phosphoenolpyruvate carboxykinase (EC4.1.1.32) activity was measured according to the methods described by Kirchner et al. (2003). The reaction system contained 2 mM nosine-5′-diphosphate disodium salt, 5 mM MnCl2, 1.1 U/mL malate dehydrogenase, 2.5 mM phosphoenolpyruvic acid monopotassium salt, 0.12 mM NADH (reduced form of nicotinamide-adenine dinucleotid) and 0.1 mol/L Tris-HCl (pH 7.4). One unit of PEPCK activity was defined as the amount of NADH generated by per g protein per minute at 30°C.
The G6Pase (EC3. 1. 3. 9) activity was measured following the methods described by Panserat et al. (2000b). The reaction system contained 26.5 mM G6P, 1.8 mM EDTA, 2 mM NAD+, 0.5 to U/mL mutarotase, 5 to 7 U/mL glucose dehydrogenase, and 100 mM imidazole-HCl. One unit of G6Pase activity was defined as the amount of enzyme that catalyzed the hydrolysis of 1 μM of glucose-6-phosphate/min under the specified conditions (30°C).
All enzyme activities were expressed per mg of total protein (specific activity). The total protein content in crude extracts was determined at 30°C using bovine serum albumin as a standard based on the method of Bradford (1976).
Real-time polymerase chain reaction measurement of hepatic growth hormone gene
We used the M. amblycephala cDNA sequences in GenBank to design the primers for GH (accession No AF463498.2) and β-actin (accession No AY170122.2) (Zhou et al., 2013). The primers were 5′-GCGGAG CCATCTCAAACAGCC-3′ and 5′-AAGCAAGCCAGAA GACGAAAG-3′ for GH and 5′-TCGTCCACCGCAAA TGCTTCTA-3′ and 5′-CCGTCACCTTCACCGTTCCA GT-3′ for β-actin. All primer pairs were synthesized by Shanghai Generay Biotech Co., Ltd. (Shanghai, China). They amplified a single polymerase chain reaction (PCR) product with the expected size as determined by the Tm value, agarose gel electrophoresis, and melting curve analysis. The PCR products were 100 to 150 bp long.
We extracted RNA from the liver tissue using RNAiso Plus (Dalian Takara Co. Ltd., China). RNA samples were treated with RQ1 RNase-Free DNase (Takara Co. Ltd., Dalian, China) to avoid genomic DNA amplification. We generated cDNA from 500 ng DNase-treated RNA using ExScript RT-PCR Kit (Takara Co. Ltd., Dalian, China). The reverse transcription PCR reaction solution consisted of 500 ng RNA, 2 μL 5×Buffer, 0.5 μL dNTP Mixture (10 mM each), 0.25 μL RNase Inhibitor (40 U/μL), 0.5 μL OligodT-adaptor primer (50 mM), 0.25 mL ExScript RTase (200 U/μL), and diethylpyrocarbonate H2O, up to a final volume of 10 μL. The reaction conditions were as follows: 37°C for 15 min, 85°C for 5 s, and 4°C thereafter.
We used real-time PCR to determine mRNA levels using an SYBR Green I fluorescence kit (Zhou et al., 2013). Real-time PCR was performed in a Mini Opticon Real-Time Detector (Bio-Rad, Hercules, CA, USA). The fluorescent quantitative PCR reaction solution consisted of 12.5 μL SYBR premix Ex Taq (2×), 0.5 μL PCR Forward Primer (10 μM), 0.5 μL PCR Reverse Primer (10 μM), 2.0 μL reverse transcript reaction mix (cDNA solution), and 9.5 μL dH2O. The reaction conditions were as follows: 95°C for 2 min, followed by 44 cycles consisting of 95°C for 10 s, 62°C for 20 s, and 72°C for 15 s. The fluorescent flux was then recorded and the reaction continued at 72°C for 3 min. We measured the dissolution rate between 65°C and 92°C. Each increase of 0.2°C was maintained for 1 s and the fluorescent flux was recorded. We calculated the relative quantification of the target gene transcript (GH) with a chosen reference gene transcript (β-actin) using the 2−ΔΔCT method. This mathematical algorithm, which does not require a calibration curve, computes an expression ratio based on real-time PCR efficiency and the crossing point deviation of the sample versus a control. We measured the PCR efficiency by constructing a standard curve using a serial dilution of cDNA; ΔΔCT = (CT, Target – CT, β-actin) time × – (CT, Target – CT, β-actin) time 0.
Calculations and statistics
where W0 and Wt are the initial and final body weights, and T is the culture period in days.
Results are presented as mean±standard error. Data were analyzed by two-way analysis of variance using the SPSS 16.0 (SPSS, IL, USA). A p-value of <0.05 indicated that there was a significant difference.
RESULTS
Growth performance and feed utilization
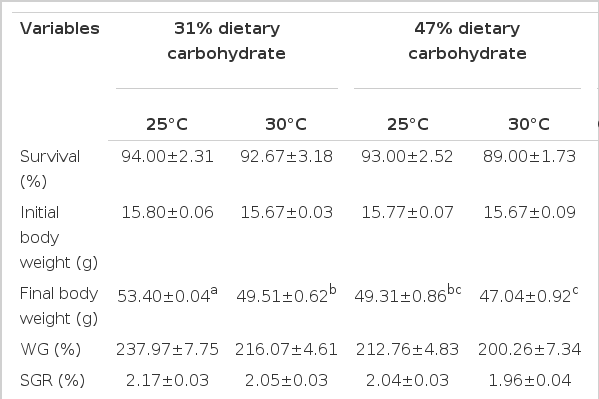
Growth performance and feed utilization of M. amblycephala are presented in Table 2. After the 8-week feeding trial, survival rates of fish were all above 89% and showed little difference (p>0.05) among all the treatments. At each temperature, final body weight, WG, SGR, and PER of fish fed the control diet were significantly higher than those fed the high-carbohydrate diet, and the FCR and HSI of the fish fed the control diet was lower than that fed the high-carbohydrate diet (p<0.05). The FR, HSI, VSI, CF and PER were not significantly different between two temperatures (p>0.05). The FR, VSI, and CF were not significantly different between diets at each temperature (p>0.05). Significant interaction between temperature and diet was found for HSI and CF (p<0.05).
Body composition
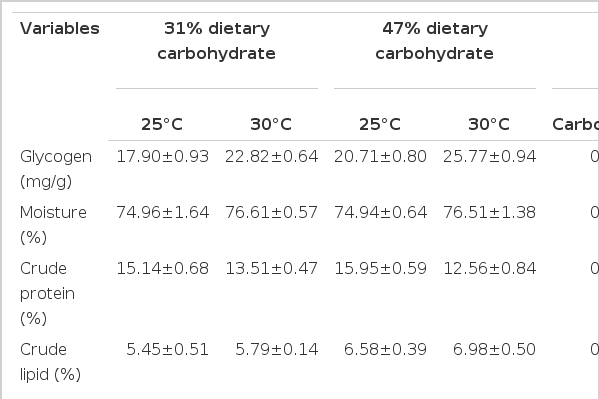
Proximate composition of whole body of M. amblycephala are shown in Table 3. Compared to those reared at 30°C, the fish reared at 25°C had lower glycogen content, higher crude protein and ash in whole body (p<0.05). At each temperature, crude lipid in whole body of the fish fed the control diet was lower than that fed the high-carbohydrate diet (p<0.05). The glycogen content, moisture, crude protein and ash in whole body were not significantly different between diets at each temperature (p>0.05). The crude lipid and moisture in whole body were not affected by temperature (p>0.05).
Serum glucose, cholesterol, and triglyceride levels
Serum glucose, cholesterol, and triglyceride levels of M. amblycephala are shown in Table 4. Compared to those reared at 25°C, the fish reared at 30°C had lower serum cholesterol and triglyceride (p<0.05). At each temperature, serum glucose of the fish fed the control diet was lower than that fed the high-carbohydrate diet (p<0.05). The serum cholesterol and triglyceride were not significantly different between diets at each temperature (p>0.05). The serum glucose was not affected by temperature (p>0.05) (Table 4).
Hepatic enzyme activities
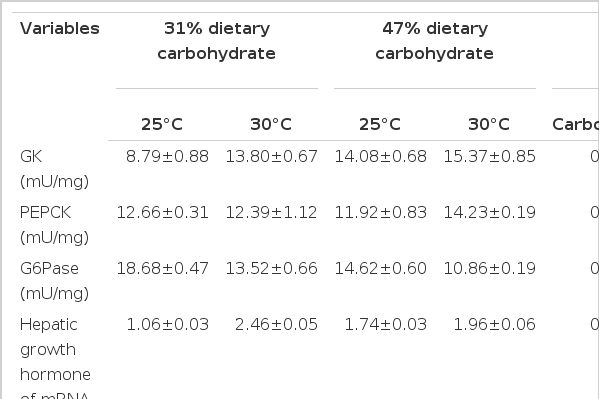
Hepatic enzyme activities of M. amblycephala are shown in Table 5. Compared to those reared at 30°C, the fish reared at 25°C had higher hepatic G-6-Pase activity (p<0.05). At each temperature, the fish fed the control diet had lower hepatic GK activity, higher hepatic G-6-Pase activities (p<0.05). The hepatic PEPCK activity was not significantly different between diets at each temperature (p>0.05). The hepatic GK and PEPCK activities were not affected by temperature (p>0.05). Significant interaction between temperature and diet was found for hepatic GK activity (p<0.05).
Hepatic growth hormone gene expression
Hepatic GH gene expression of M. amblycephala are also shown in Table 5. Compared to that reared at 25°C, the fish reared at 30°C had higher relative levels of hepatic GH gene expression (p<0.05). The relative levels of hepatic GH gene expression were not significantly different between diets at each temperature (p>0.05). Significant interaction between temperature and diet was found for the relative levels of hepatic GH gene expression (p<0.05) (Table 5).
DISCUSSION
Several previous studies have showed that incorporation of appropriate levels of dietary carbohydrates in fish feeds can improve their growth performance and provide an inexpensive non-nitrogenous energy source (Hemre and Hansen, 1998; Peragón et al., 1999; Vielma et al., 2003). However, excess levels of carbohydrates reduce the growth rate and feed utilization (Vielma et al., 2003; Enes et al., 2006). In this study, we found depressed growth and feed utilization in M. amblycephala fed diets high in carbohydrate content. The finding of our study indicated that high dietary carbohydrate may reduce the growth rate and feed utilization in M. amblycephala, similar to previous studies (Furuichi and Yone, 1980; Tan et al., 2009; Miao et al., 2011).
In general, the increase of HSI that accompanied the increasing dietary carbohydrate level may be related to the increased glycogen deposition in the liver of fish fed high-carbohydrate diets (Wilson, 1994; Moreira et al., 2008). In this study, contrary to the CF, HSI were increased when the two factors; high-carbohydrate diet and higher temperature treatment co-existed. HSI and whole body lipid content was significantly affected by the dietary carbohydrate level in the current study, as in European sea bass (Moreira et al., 2008). In our study, we found that glycogen content, crude protein and ash in whole body were affected by temperature suggesting that temperature influences the physiological characteristics of ectotherms (Gillooly et al., 2001; Hochachka and Somero, 2002; Clarke, 2004) and it also modulates utilization of nutrients by fish (Keembiyehetty and Wilson, 1998).
Serum biochemical parameters played an important role in evaluating the health of many organisms (Abdel-Tawwab et al., 2010). It is generally accepted that fish have limited ability to metabolize glucose, and high digestible carbohydrate intake results in postprandial hyperglycemia for many hours (Wilson, 1994). In our study, we found that M. amblycephala fed high-carbohydrate diet had higher serum glucose levels. A similar positive correlation between blood glucose concentration and dietary carbohydrate level was found in cobia, Rachycentron canadum L. (Ren et al., 2011). In this study, we found that M. amblycephala reared at 30°C had lower serum cholesterol and triglyceride, similar to a previous study on crucian carp (Carassius auratus) (Han et al., 2010).
The change of carbohydrate content in the fish diet induces pronounced changes in carbohydrate metabolism, especially in the activity of key enzymes of carbohydrate metabolism in the liver (Leung and Woo, 2012). Previous studies reported that GK activity increased with the increase in dietary carbohydrate in rainbow trout (Oncorhynchus mykiss) and gilthead sea bream (Sparus aurata) (Panserat et al., 2000b; Metón et al., 2004). The G6Pase activity decreased with the increased dietary carbohydrate level in common carp (Cyprinus carpio) and gilthead seabream (Sparus aurata) (Panserat et al., 2002). In our study, hepatic GK activity of M. amblycephala was increased greatly by the application of both a high-carbohydrate diet and a higher temperature. In addition, we found that temperature negatively modulated hepatic G-6-Pase activity. Thus, both dietary composition and temperature seemed to be the important factors controlling hepatic key metabolic enzymes involved in carbohydrate metabolism in Wuchang bream.
Growth hormone is synthesized in the pituitary gland and secreted into the bloodstream under the regulation of neuronal, hormonal, and nutritional factors (Duan, 1998). In fish, GH can participate in protein, fat, and carbohydrate metabolism (Donaldson et al., 1979). Growth hormone mRNA was detected in the pituitary gland, brain, gill, heart, kidney, muscle, and liver of common carp (Kumar et al., 2013) and rainbow trout and coho salmon (Tymchuk et al., 2009). The content of CH is influenced by environmental factors such as temperature and light regime (Hemre et al., 2002). In this study, the relative level of hepatic GH gene expression was increased greatly in fish subjected to the high-carbohydrate diet and higher temperature treatment. Our results were also in agreement with a previous study showing that there was an overall negative correlation between expression of the GH gene and growth performance for all common carp diet groups (Kumar et al., 2013). This finding indicated that high temperature may lead to metabolic stress in M. amblycephala. This could be explained that the GH levels are elevated in the metabolic disorders that shared an increased catabolism/anabolism balance (Pérez-Sánchez and Le Bail, 1999). However, the relationship between temperature for M. amblycephala and GH remains to be further investigated.
CONCLUSION
In conclusion, the present study suggested that HSI, CF, hepatic GK activity and the relative levels of hepatic GH gene expression of M. amblycephala were affected greatly by the interaction between the two factors of high-carbohydrate diet and higher temperature treatment.
ACKNOWLEDGMENTS
This work was supported by the National Natural Science Foundation of China (31472289); Open Fund of Key Laboratory of Freshwater Fisheries and Germplasm Resources Utilization of Ministry of Agriculture (KF201311); the National Non-profit Institute Research Grant of Freshwater Fisheries Research Center, Chinese Academy of Fishery Sciences (2013JBFR03); the National Technology System for Conventional Freshwater Fish Industries, the Modern Agriculture Industrial Technology System (CARS-46); the Special Fund for Agro-Scientific Research in the Public Interest (201003020); Special Scientific Research Funds for Central Non-profit Institutes, South China Sea Fisheries Research Institute, Chinese Academy of Fishery Sciences (2014YD01).